Although modern medicine is effective in fighting disease, their full potential is often not recognized because about 50% of patients do not take their medications as prescribed. Factors contributing to poor medication adherence are endless and encompass those that are related to patients (e.g., negligible knowledge on health literacy and insignificant involvement of the patient in the medical treatment decision process). Those that are related to physicians (e.g., The prescribe complicated drug regimen, with not much follow up creating a communication gap and patient uninformed about side effects of the medication). causes numerous problems such as Drug overdose and medicine noncompliance that has dramatically impacted the United States over the last decade. In 2016 marked the highest deaths related to drug overdose, as well as the paramount economic blow of over 300 billion dollars spent on patient noncompliance and medication misuse. While smart bottles have been developed to simplify dose tracking and medication taking, the current designs in the market for preventing drug sharing and noncompliance are expensive or extremely consumer-unfriendly.
Projects
Since I started my studies in Chemistry, I have been devoting time to doing outreach.
BLODOT
Impoverished residents often encounter challenges limiting their healthcare, including shortages of healthcare professionals, essential medical technology, and sterile facilities. This further drives sickness within underdeveloped countries. A complete blood count, or CBC, test is generally the first step in the diagnosis. It measures many blood parameters and provides the physician with information that would narrow the possible illnesses. The machinery involved in a CBC test can be challenging to implement in underdeveloped populations. The size, costs, and overall requirements for its placement make it unfit to function in such environments.
The development of a CBC point-of-care (POC) device would effectively replicate large-scale CBC machinery’s function and quickly give patients results. Through microfluidics, the device will separate whole blood into red blood cells (RBCs), white blood cells (WBCs), and platelets. Dielectrophoretic separation methods utilize electrodes within the chip’s main channel to further separate these blood cells into individual channels specific to CBC parameters that will later be counted via flow cytometry.
The handheld device would not weigh more than 2-3 lbs, with dimensions around 160mm x 100 mm x 50mm. The device should produce efficient results within 2-4 min. Estimations show that the device would be able to present products with 90-100% accuracy. Currently, the device is at the paper-prototype level, focusing on implementing the critical parameters of the CBC test in the device. Work towards a wireframe prototype, estimated to be finished in August 2019, adding more panels and features to this device.
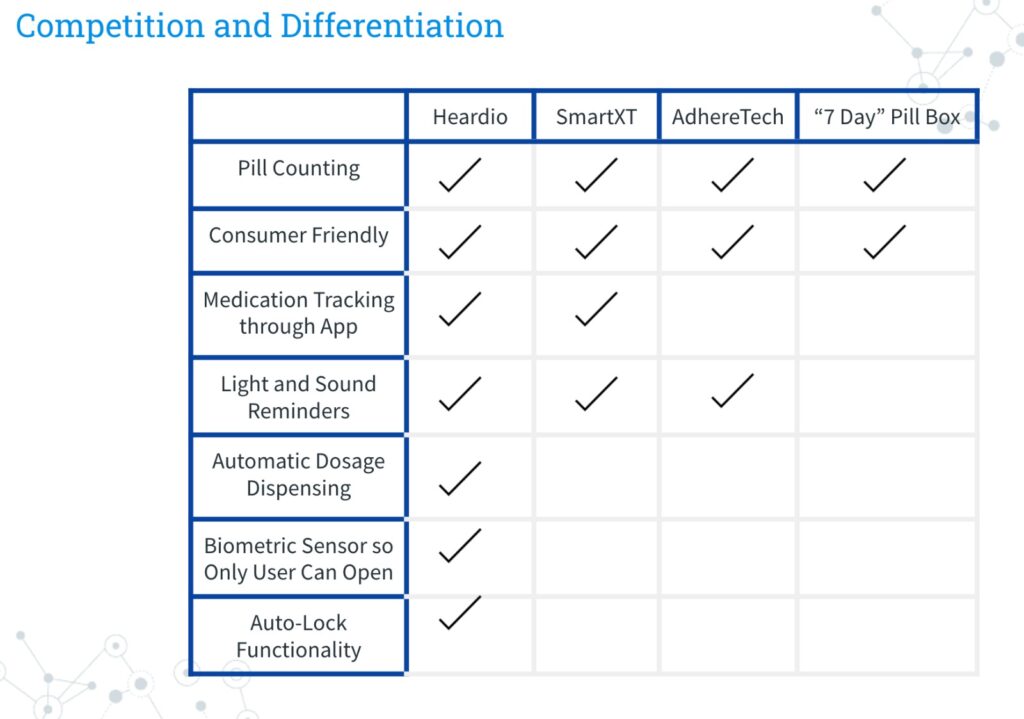
HEARDIO
Value Proposition:
Medical Industry has generally used a bleak solution to ensure that this adherence issue is dealt with by informing the patient when they pick up the medication about how he or she is supposed to consume the medication and what will be the side effects of the patient not adhering to the medication. But in spite of this, the problem remains unsolved. The patients still end up missing 50-60% of their drugs. The Problem remains unresolved.
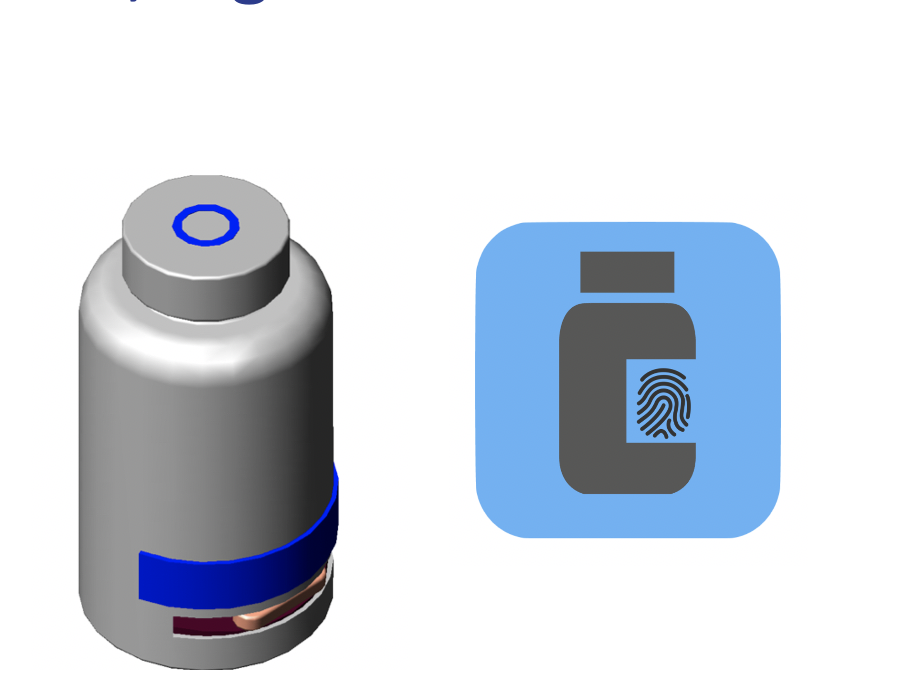
My team of engineers and scientists have come up with a non-traditional solution. Unlike any other, solution Heardio is going to bring to the market a revolutionary medical pill dispenser that dispenses the right dosage to the right person at the right time. Unlike, other medical pill bottles and solutions available in the market that only focus on reminding the user about taking the medication. This new recyclable innovation ensures that no one but the user tampers with the medication. It also keeps giving a reminder to the user who makes the drug. The dispenser is connected to a mobile application on your smartphone, to remind you as well as assist the user in taking the medicines via using the fingerprint sensor located on the smartphone, can either be used on the cell phone or the bottle itself to unlock the container, making sure no one but the user can use the medication. Heardio will take taking medical adherence to the last pill level.
This amazing technology will work toward minimizing the giant adherence loophole we have in any line of treatment, as consistency is the key to success.

HEARDIO AND IT’S APP LOGO
The Solution:
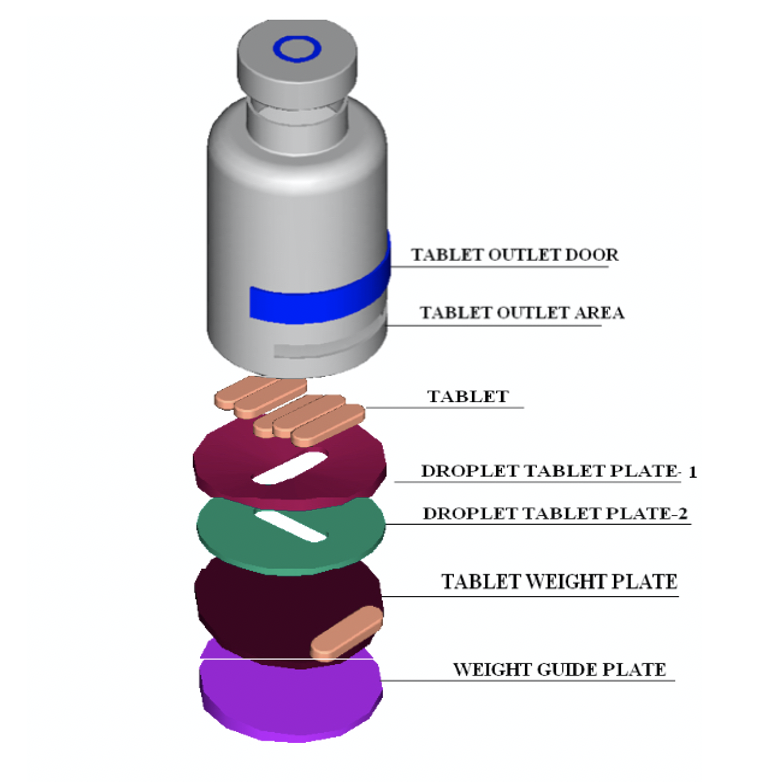
Heardio is a medical pill dispensing system consisting of a smart bottle and mobile app to allow patients to receive the correct dosage at the right time. The unique “one-dose” dispensing mechanism and auto-lock functionality of the bottle, paired with medication tracking features of the app, allows for total medication adherence.
Market:
Based on the facts our current market focus is enhancing medication adherence in a niche market of post-surgery, heart failure geriatric population. This market is about 300K. As of now but we plan to diversify into medication adherence in the future.
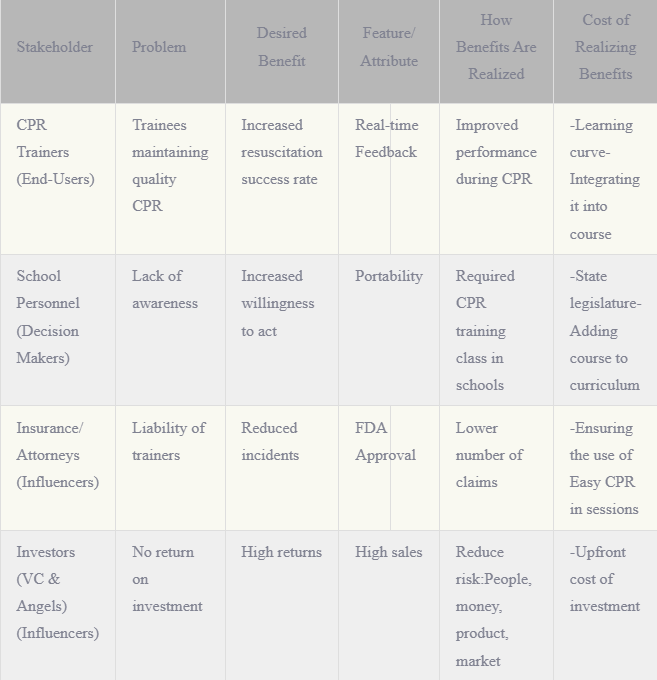
Stakeholders:
- Patient & Family
- Consumers directly affected by medication adherence issues.
- Pharmaceutical companies
- Increase in medication adherence will increase sales
- Pharmacies
- Have knowledge in proper adherence critical care medications to aid their patients
- Hospital:
- Increase in adherence will decrease patient rehospitalization, decreasing hospital costs and penalties associated with readmittance.
My Contribution: I developed and engineered the pill enclosure for critical care patients for better medication adherence, by discussing the product design with a team of engineers, focusing on design criteria to better suit patient’s needs. In order to complete this project, I collaborate with a team of Entrepreneurs from the Healthcare and Pharmacy Business Management field for the commercialization of the product. I was mentored by Mr. Pritam Advani.
I also performed customer discovery, created a business and financial model for a startup as well as developed the Auto Cad models of the project. Had regular product progress presentations and final investors pitch.
CPR ASSISTANT
CPR assist reduces discomfort that comes with administering CPR by providing an assistive device used in training and real-world application. CPR-assist ensures that proper depth and compression rate is achieved while performing CPR.. As well as have the training fresh in trainees’ heads.

Prevent Self Harm in patients with Autism Spectrum Disorder
Problem Description
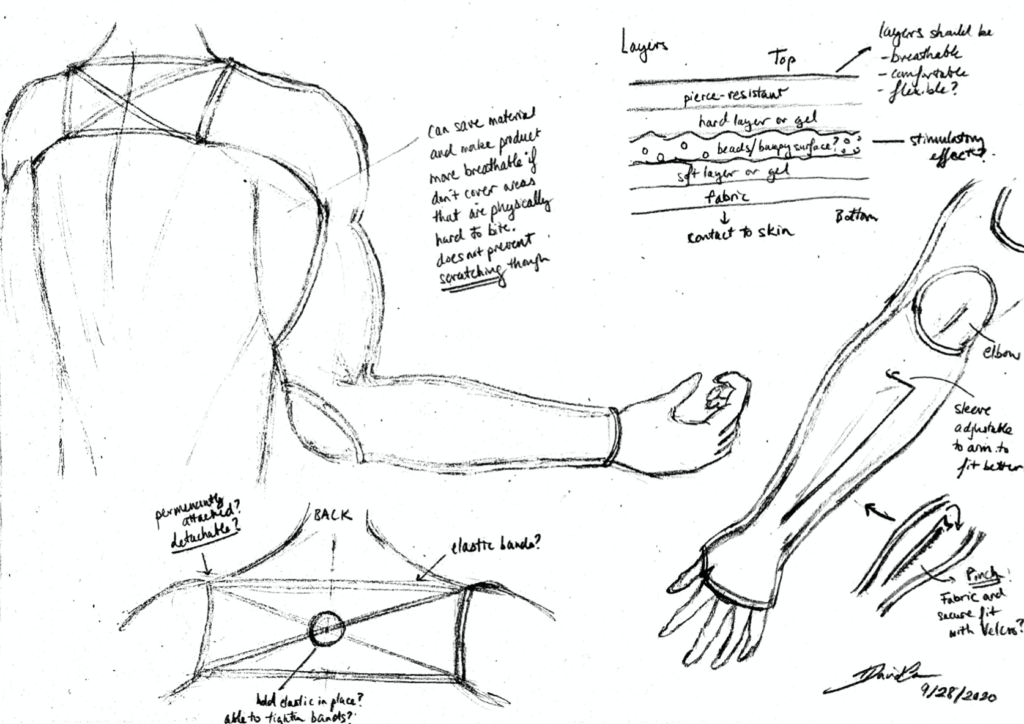
When patients with Autism Spectrum Disorder (ASD) and other developmental disorders are agitated or in a panicked state, many end up harming themselves by biting or scratching their arms as a coping mechanism. Parents of children with these conditions struggle to prevent this behavior as they get older and physically stronger. Current devices to prevent biting do not protect the full-arm or are simply not feasible to wear for prolonged periods. We believe that finding a way to prevent these patients from self-harming while still being able to bite and scratch themselves as a coping mechanism is a serious need that should be addressed.
My Contribution
As a part of the team, I worked with the FDA quality system regulation (QSR) with emphasis on the Design Controls (21 CFR 820.30). The Specific experience includes ethnography and human factor analysis, formulation of technical specifications, failure mode, and effects analysis (FMEA), verification and validation (V&V) planning. One of the interesting Verification protocols I developed the drying rate protocol. to test the drying of the sleeve we designed.
I also gained experience Experience with the formulation of a comprehensive Design History File (DHF).

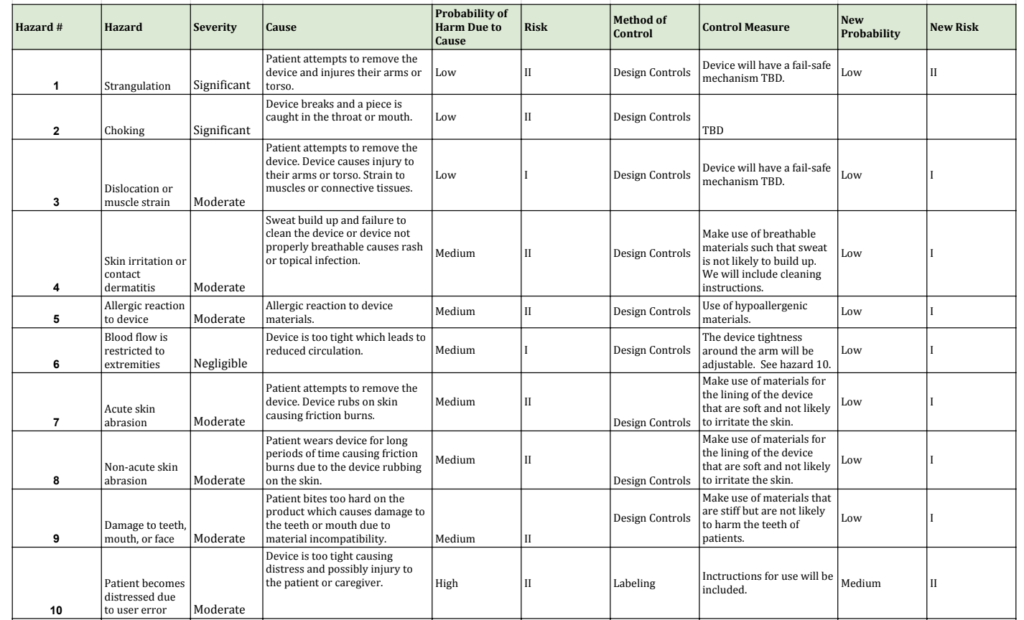
Hazard and Risk Analysis:
Hazards and risks were identified by looking at known risks of existing designs and hazards associated with protective equipment. Ten hazards were identified, each with quantifiable severity, potential causes, probability of harm due to cause, risk, possible control measures, and adjusted risk. Identified hazards were: strangulation, choking, dislocation or muscle strain, skin irritation or contact dermatitis, allergic reactions, blood flow restriction to extremities, acute skin abrasion, non-acute skin abrasion, damage to teeth, mouth, or face, and patient injures themselves or caretaker due to emotional distress.
Based on the hazard’s risk and probability of severity, changes to the design were made to ensure a better and safer product. Changes included using a tighter woven material as the outer layer, industrial stitching at seams, and using elastic straps.
Final Design:
The current design is a sleeve made of two layers of Kevlar, a neoprene and silicone rubber middle layer for added protection, and an inner layer of a cotton/spandex blend fabric. Sleeves were designed to have an elbow hole and a thumb hole. Both sleeves are connected by an elastic strap with a buckle on the back. Figure 1A features the material composition and arrangement of the device. Figure 1B and Figure 1C show the back and wrist securement respectivefully.

Verification and Validation Results:
Four verification tests were performed to assess the device’s ability to withstand baseline safety and effectiveness for use. The tests performed were Puncture Assessment, Machine Washability Assessment, Abrasion Resistance Assessment, and Durability Assessment. Results from each verification study are summarized as follows:
- Puncture Assessment: The device can withstand high forces and high pressures that are above the range of an average bite.
- Machine Washibility Assessment: The device should be washed by hand only. The device should be left to air dry for at least 12 hours after washing.
- Abrasion Resistance Assessment: Both layers of Kevlar were fully abraded at extreme conditions. However, the device could be expected to not deteriorate from daily wear and tear easily.
- Durability Assessment: The first layer of Kevlar was fully abraded by the end of the first or second run. The device failed the acceptance criteria, but the protocol needs to be adjusted to accurately represent everyday wear and tear.
In summary, verification studies revealed that the device is highly protective from puncture and abrasion, but fails to be machine washable. While the device needs modifications to the design to better meet user needs, validation studies revealed that there is a general satisfaction with the device. Slight concerns were identified regarding the breathability and thickness of the device, but responses showed high satisfaction with the protective abilities of the sleeves. Overall, the device is, at best, satisfactory to customer needs, but needs modifications to the design and manufacturing process so that the device
Unmet Need Identification:
In early September, the first steps our team took in identifying unmet clinical needs was looking into our own experiences and the different problems we have encountered. A list of roughly 15 ideas for clinical unmet needs was identified. We then reached out to individuals who may be more knowledgeable or experienced in the issues we were trying to address. On September 25, Shawn Danhouser, a parent and a soccer coach for children with special needs, provided us with feedback that helped us identify the need for protective equipment to prevent self-injurious behavior in individuals with Autism Spectrum Disorder (ASD).
After identifying this unmet need, we developed a survey inquiring about self-injurious behavior in patients with ASD. Questions included frequency of biting, severity of injuries, if they use any protective devices, and any anecdotal experiences they were willing to share. From our survey, 35% of the responses said that they know someone who bites or scratches themselves as a coping mechanism, 50% of which leave open wounds. The majority of surveyors said that there is a medium to high frequency of self-harming behavior. This preliminary ethnographic data validated Shawn Danhouser’s claim of this unmet clinical need as a widespread issue. Therefore, we decided to pursue this as the foundation of our Senior Design project.
Design Inputs:
Through the identification of user needs, the killer experiments, and input from experts, we defined the following design inputs:
- Size: The device should have an upper arm length adjustable between 20-40 cm and an adjustable circumference.
- Maintenance and Cleaning: Washable using a machine washer and dryer. Can be washed with detergent and warm water
- Materials:
- Outer layer: Kevlar
- Middle layer: Neoprene and silicone rubber
- Inner Layer: Spandex fabric
- Fastening mechanism: plastic lock with elastic strap
- Should not have materials that cause skin irritation and allergic reactions; no sharp projections.
- Weight: 2 pounds maximum
- Product Life: 3-5 years
- Packaging: Our device will be sealed in plastic film and shipped in a cardboard box with a volume less than 12 inches cubed.
- Labeling: Should be on the outside of the product using a straight cut/flat label or loop fold. Should contain following information: size, care/cleaning instructions, material information, brand name.
- Usability: Device should be slipped on in less than a minute by parent/caregiver.
- Durability: Device should be able to protect from puncture and pressure wounds caused by a pressure of 1.75 Newtons/cm2 from the front incisors.
Natural Language Processing Project
Project is Cooking "Wait for it to come!!"
Design is a process. As of now, I am working on developing this project.

